*This is an article from the Spring 2022 issue of Contentment Magazine.
By Lewis S. Coleman, MD, FAIS
“No great discovery was ever made without a bold guess.” –Isaac Newton
Introduction
For thousands of years, physicians focused their efforts on facilitating the mysterious healing property present in humans and animals. This changed during the past 200 years after germ theory and cell theory introduced the notion that diseases are distinctly different from one another, so that individual diseases must be diagnosed and cured using specialized treatments. This seems reasonable in the case of infectious diseases, which often produce distinctive symptoms and can be prevented with specific vaccines and cured with specialized treatments. However, modern medical theory is plagued by numerous shortcomings. It cannot explain the nature of disease, or why seemingly disparate diseases occur in concert. For example, diabetes, hypertension, obesity, and cancer are closely related. Nor can conventional theory explain why most diseases manifest fluctuating common symptoms such as fever, fatigue, malaise, cachexia, inflammation, exudates, and rashes. Furthermore, it cannot adequately explain embryological development or the everyday manifestations and relationships of hemodynamic physiology, tissue repair and stress, such as blood pressure, pulse rate, cardiac output, and so forth.
During WWII Dr. Hans Selye postulated that an undiscovered “stress mechanism” explains the nature of disease.1-4 This offered the simplest, and therefore the most scientifically promising prospect for an effective theory of medicine, as science seeks the simplest explanation of disparate phenomena, and it represented a reversion to the traditional medical viewpoint, which acknowledges the ability of the body to heal itself without medical help.
The discovery of DNA focused attention on Selye’s ideas, because although DNA explains the retention and replication of genetic characteristics, it cannot by itself explain the nature and relationships of physiology, disease, and stress. Medical researchers therefore hypothesized that Selye’s stress mechanism works closely with DNA during embryological development (i.e., pregnancy) to convert the genetic blueprint into multicellular structures. They further hypothesized that the stress mechanism remains active for the duration of life to repair tissues and regulate organ function, while DNA itself resumes quiescence. These powerful ideas inspired an intense international search for the stress mechanism that lasted 30 years and consumed the careers of hundreds of researchers, the lives of thousands of tortured test animals, and millions (today billions) of dollars. However, even during the heyday of stress research, most researchers doubted that a single mechanism could explain both tissue repair and organ regulation. Therefore, some presumed that the stress mechanism governs the orderly sequence of tissue repair, 5 while others embraced capillary gate theory, which presumed that a submicroscopic molecular mechanism governs capillary flow and organ function. 6 Unfortunately, neither approach was able to identify any testable mechanism that might explain any aspect of Selye’s concept, and his ideas were relegated to the realm of the Unicorn.
Most powerful theories arrive many years before accumulating evidence from unrelated research provides the evidence necessary to test and confirm them, and that was the fate of stress theory. Even now, 50 years after Selye’s ideas were abandoned, it seems astonishing that the newly discovered stress mechanism can explain embryology, physiology, pathology, stress, and their relationships. Furthermore, it exceeds the bounds of medicine and confers a unified theory of biology, with implications that reside in the realm of science fiction. However, this essay will confine itself to a simplified presentation of the stress mechanism that is intended to facilitate future essays that will explore its operation and implications in greater detail.
Cell Biology
There are three known domains of independent, free-living cells: bacteria, Archaea, and Eukarya, but complex multicellular animals and plants are composed exclusively of eukaryotic cells. The reason is that Bacteria and Archaea respire (generate energy) via their cell walls, which limits them to small size, limited shapes, and solitary existence. They thrive in vast multitudes in the intense heat far below the earth’s surface. They have evolved numerous metabolic pathways that enable them to thrive on toxic chemicals, sewage, and even pure electricity, and their combined activities produce water, oil, and atmospheric gases. However, their cell walls must be exposed to the environment to enable them to respire, so that they are incapable of forming complex multicellular plants and animals.
In contrast, Eukaryotic cells generate their energy from within via a metabolic pathway called the “Krebs cycle” that consumes glucose and oxygen, and produces water and carbon dioxide in the process of generating an energetic chemical called adenosine tri-phosphate, or ATP, that energizes all their cell functions. The Krebs cycle exists only within “organelles” called mitochondria that are contained in the cytoplasm of eukaryotic cells. Their DNA is isolated from the chromosomal DNA that determines genetic inheritance by the thick walls of the cell nucleus. The mitochondria are believed to be the remnants of previously free-living bacteria that were ingested by eukaryotic cells in the distant past, and somehow became incorporated into the eukaryotic cell.
Because eukaryotic cells generate ATP energy from within, they are far more capable and complex than bacteria and Archaea. They can differentiate, de-differentiate, and re-differentiate into specialized cells to form the tissues and organs of complex multicellular plants and animals. Although the eukaryotic cells of complex multicellular animals and plants generate their ATP energy from within using the Krebs cycle, they must be continuously supplied with glucose and oxygen, and the waste products of food digestion and organ activity must be removed from the body. That is the function of the stress mechanism, which repairs tissues, regulates organ function, and otherwise maintains the “internal milieu” (internal environment) of mammals.
The Stress Mechanism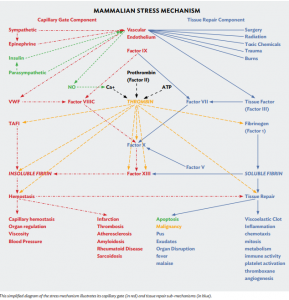
The stress mechanism appears early during embryological development. It converts the DNA genetic blueprint into complex multicellular structures during pregnancy, and remains active for the duration of life, while chromosomal DNA resumes quiescence once embryological development is complete.7
The stress mechanism can best be comprehended as consisting of two sub-mechanisms that share the interaction of blood enzyme factors VII, VIII, IX and X, so that they consume the same substrates and produce the same products (thrombin, soluble fibrin, and insoluble fibrin).
Figure 1. This simplified diagram of the stress mechanism illustrates its capillary gate (in red) and tissue repair sub-mechanisms (in blue).
The tissue repair sub-mechanism activates factor VII in accord with tissue damage to repair tissues, and the capillary gate mechanism activates factor VIII in accord with nervous activity to regulate cardiac output, blood pressure, pulse rate, tissue perfusion, tissue oxygenation, and organ function. The activity of each sub-mechanism exaggerates that of the other, which focuses the activity of the stress mechanism to repair tissues and regulate organ function. The independently fluctuating activities of the two sub-mechanisms produces a baffling blizzard of symptoms and manifestations that obscure the relatively simple function of the mechanism.
Stress mechanism hyperactivity induced by excessive and unremitting combinations of environmental stresses causes the stress mechanism to consume and waste its substrates and produce excessive and defective quantities of its products that disrupt organ function and damage tissues. This manifests as disease.
The Enzymatic Engine of the Stress Mechanism
The liver continuously produces fibrinogen and enzyme factors VII, VIII, IX, and X, and releases them into flowing blood. The enzymatic interaction of factors VII, VIII, IX and X generates thrombin, converts fibrinogen to soluble fibrin, and converts soluble fibrin to insoluble fibrin. These are the three basic products produced by the stress mechanism.
I call thrombin the “universal enzyme of energy transformation.” It converts ATP energy into the cell and extracellular enzyme activities of the stress mechanism. It is analogous to intracellular ATPase enzyme that converts ATP into intracellular functions. Both require ATP and Ca+. The parathyroid glands maintain blood Ca+ levels within a narrow range to optimize extracellular thrombin activity.
Thrombin energizes the conversion of fibrinogen to soluble fibrin, and the conversion of soluble fibrin to insoluble fibrin. It energizes gelsolin, which digests actin that escapes into blood, and TAFI (thrombin-activated fibrinolysis inhibitor), which stabilizes blood clots. It energizes cellular mitosis, platelet activation, and cellular metabolism, specialization, hypertrophy, chemotaxis, hormone release, immune activity, and angiogenesis to enable tissue repair. Abnormal thrombin elevations cause systemic inflammation that disrupts organ function and exaggerates cell proliferation, causing repair cells to invade and disrupt adjacent tissues, stimulate nervous activity, and release tissue factor. Such abnormal activity causes tumor formation that sometimes becomes malignant (i.e., self-sustaining).
I call soluble fibrin the “universal substrate of tissue protein and tissue repair.” Its fundamental significance is overlooked in medical literature. It is a fibrillar (string-like) protein that infiltrates inflamed tissues and forms a protein matrix in damaged tissues that facilitates the formation of granulation (repair) tissue that fills empty spaces and binds damaged tissues together. It is also a substrate of collagen, elastin, insoluble fibrin, pus, exudates, mucus, saliva, lipoproteins, and milk. When produced in excess, it causes tissue edema, disrupts organ function, and promotes sclerosis that threatens permanent tissue and organ damage.
I call insoluble fibrin is the “universal polymer of hemostasis.” It is generated within capillaries in accord with sympathetic nervous activity to “close” the capillary gate mechanism by increasing capillary flow resistance. In the event of tissue damage, insoluble fibrin entangles blood cells and platelets to enable capillary hemostasis and clot formation that stems blood loss. Excessive insoluble fibrin generation threatens hypercoagulability and tissue ischemia that manifests as myocardial infarction, strokes, and pulmonary embolus.
The Vascular Endothelium
The vascular endothelium mediates the activities of both the capillary gate mechanism and the tissue repair mechanism. It is a delicate layer of highly specialized cells, one cell thick, that lines the inner surface of all blood vessels and is the sole constituent of capillaries, which are the smallest blood vessels in the body. Thus, it is ubiquitous throughout the body. Its cells react to local circumstances and communicate with one another via electrical signals. It produces the glycocalyx, a diaphanous molecular matrix that lines the inner surface of blood vessels and regulates fluid absorption.
The vascular endothelium isolates blood enzymes from tissue factor in extravascular tissues, but it is “selectively permeable.” For example, it allows tissue factor to escape from extravascular tissues into flowing blood, and it allows factor VII to penetrate slowly from blood into extravascular tissues, but is impermeable to Factors VIII, IX, and X. It is specialized to facilitate the function of organs and tissues. For example, its cells are tightly joined in the brain, which explains why medications and toxic substances do not readily penetrate brain tissue. This is called the “blood-brain barrier.” In contrast, it is relatively permeable in the kidney and liver to facilitate urine production and neutralize toxic substances in the digestive system.
Tissue damage disrupts the vascular endothelium and directly exposes blood enzymes to tissue factor in the damaged tissues and activates the tissue repair mechanism. Inflammation increases the permeability of the vascular endothelium and invites harmful exposure of tissue factor to blood enzymes.
The autonomic (automatic) nervous system innervates the vascular endothelium and causes it to release hormones (nitric oxide and von Willebrand Factor) that govern the capillary gate mechanism.
The Tissue Repair Sub-Mechanism
The tissue repair sub-mechanism repairs tissues in accord with tissue disruption. It consists of tissue factor that is produced by extravascular cells, the interaction of enzymatic factors VII, VIII, IX, and X, and the vascular endothelium that isolates blood enzymes from tissue factor.
Tissue factor stabilizes and thereby activates blood enzyme factor VII, which by itself is so labile that it cannot express its enzymatic activities. Factor VII activity is essential to enable the function of factors VIII, IX, and X, so that factor VII functions as a “trigger” that determines the location and magnitude of coagulation, capillary hemostasis, and tissue repair. The selectively permeable vascular endothelium normally allows slow “leakage” of tissue factor into flowing blood to enable continuous low-level activity of the enzymatic interaction. It simultaneously allows slow “penetration” of factor VII into extravascular tissues, where tissue factor activates it to generate small amounts of thrombin that energize tissue maintenance.
Damage and disruption of the vascular endothelium increases the exposure of tissue factor to factor VII and activates the enzymatic interaction, which generates thrombin, soluble fibrin, and insoluble fibrin in the immediate vicinity of tissue damage. Thrombin activates platelets, causing them to become “sticky” so that they bind together to form a “white clot” that prevents blood loss. Factor VIII generates insoluble fibrin that binds red cells together into a more permanent viscoelastic “red clot” that substitutes for the vascular endothelium. Like the vascular endothelium, the red clot is “selectively permeable.” It allows the passage of factors VII and X from blood into the damaged tissues beneath its protective surface, but it remains impermeable to factor VIII due to its gigantic molecular size.
The viscoelastic clot then regulates the exposure of factors VII and X to tissue factor to generate thrombin to energize tissue repair but limit thrombin generation within safe levels to prevent malignancy (self-sustaining tissue repair activity that invades and disrupts adjacent tissues and forms tumors). Tissue repair then proceeds in an orderly and predictable sequence, beginning with inflammation that loosens cell connections to enable fibroblast repair cells to migrate from adjacent undamaged tissues into the injured tissues via chemotaxis to begin cellular tissue repair activities. Thrombin elevations energize fibroblast mitosis, multiplication, and collagen production to form granulation tissue that fills empty spaces and binds damaged tissues together. Angiogenesis provides perfusion to proliferating repair cells. Thrombin energizes immune cell activity that inhibits infection and removes debris from the damaged tissues.
Repair cells communicate with one another by subtle electromagnetic signals and by releasing chemokines, cytokines, prostaglandins, and other cell hormones to differentiate, specialize, and replace damaged tissues. As the tissue repair activity restores the integrity of the vascular endothelium, thrombin generation declines, which causes apoptosis (programmed cell death) that draws wound edges together to complete the repair process.
The Capillary Gate Sub-Mechanism
The capillary gate mechanism consists of the vascular endothelium, the autonomic nervous system, and the enzymatic interaction of factors VII, VIII, IX, and X.
Carbon dioxide is the primary regulator of the capillary gate. It directly releases nitric oxide (NO) from the vascular endothelium. NO binds to Ca+ to inhibit thrombin activity, which undermines the stabilization of insoluble fibrin by TAFI (thrombin-activated fibrinolysis inhibitor). This accelerates the disintegration of insoluble fibrin to promote capillary perfusion and improve oxygen delivery. For example, exercising muscle increases CO2 production, opens the capillary gate, and increases oxygen delivery to the exercising muscle. CO2 also releases oxygen from the hemoglobin molecule via the Bohr effect to enable cellular oxygen uptake.
Sympathetic nervous activity closes the capillary gate. It releases von Willebrand Factor (VWF) from the vascular endothelium into blood, which activates factor VIII that accelerates thrombin generation to energize its conversion of soluble fibrin to insoluble fibrin that increases flow resistance in capillaries. The increased microvascular flow resistance reduces organ perfusion and oxygenation, which inhibits organ function. It also reduces cardiac output and venous return to the heart.
Parasympathetic nervous activity releases NO from the vascular endothelium to promote organ perfusion and increase organ activity. This explains how autonomic balance governs organ activity.
Insulin and epinephrine are opposing hormones that govern the capillary gate in peripheral tissues where direct autonomic innervation is lacking. Sympathetic nervous activity releases epinephrine from the adrenal gland, which releases VWF from the vascular endothelium to activate factor VIII, generate insoluble fibrin, and close the capillary gate. Parasympathetic nervous activity releases insulin from the pancreas, which releases NO from the vascular endothelium to open the capillary gate.
Critical brain perfusion is sustained by astrocytes that release TPA (tissue plasminogen activator) that disintegrates insoluble fibrin generation to prevent capillary gate closure that threatens brain ischemia. This explains why massive doses of epinephrine during cardiopulmonary resuscitation does not halt oxygen delivery to the brain.
The Mechanism of Oxygen Transport and Delivery
Carbon dioxide is vilified as the cause of climate change and routinely regarded as “toxic waste, like urine,” but the truth is ignored. Carbon dioxide it is as essential for animal life as oxygen, and it has powerful therapeutic properties, because it optimizes the mechanism of oxygen transport and delivery that delivers oxygen to cells located deep within the body. Breathing air enriched with carbon dioxide increases tissue oxygenation. Carbogen (generally 5% oxygen mixed with 95% oxygen in a pressurized tank) is perhaps the most potent, powerful, practical, and affordable medical treatment ever discovered. It is an ideal emergency treatment for heart attacks, strokes, smoke inhalation, carbon monoxide poisoning, asthma, atelectasis, pneumonia drowning, and newborn babies with breathing problems. It was widely accepted, installed on fire trucks, and saved numerous lives in the 1930s, but was forgotten courtesy of perverse medical politics.8-11
Eukaryotic cells continuously produce carbon dioxide as a by-product of cellular respiration. It is as benign, inert, and as free of toxicity as water, but like water it can cause lethal asphyxiation or drowning,16 even though it causes a warning sense of suffocation when it is inhaled in dangerous concentrations.12,13 It readily dissolves in body fluids and tissues, so that the adult human body contains about 20 liters of CO2. By comparison, the body contains only 1 liter of oxygen and 1 liter of nitrogen. The oxygen is mostly bound to hemoglobin and continuously consumed by cells, so that disruption of the mechanism of oxygen transport and delivery causes critical oxygen starvation within minutes. The CO2 continuously escapes through the skin and equilibrates with atmospheric CO2, which slowly fluctuates over eons. Respiratory drive mechanisms adapt to this equilibrium and seek to sustain it.17
Unique among atmospheric gases, carbon dioxide inhibits the binding of oxygen to the hemoglobin molecule. This explains its pivotal role in oxygen transport and delivery. Carbon dioxide stimulates breathing, which simultaneously reduces the concentration of carbon dioxide to 5% and replenishes oxygen in the lung. This optimizes the binding of hemoglobin to oxygen as blood passes through the lung, so that all four hemoglobin binding sites are normally 100% saturated with oxygen as blood transits the lung. Thus, breathing 100% oxygen confers negligible benefit, because hemoglobin transports almost all oxygen in blood. The heart propels oxygenated blood to organs and peripheral tissues, where higher CO2 concentrations release oxygen from the hemoglobin molecule via the Bohr effect into surrounding tissues, where cells consume it.
As noted above, CO2 is the primary regulator of the capillary gate, so that rising tissue levels of CO2 in muscle tissues during exercise has the immediate effect of reducing microvascular flow resistance, which speeds oxygen transport and increases oxygen delivery to satisfy the oxygen requirements of the exercising cells.
Exercise training induces angiogenesis (capillary proliferation) in the affected muscles. This lowers flow resistance so that trained athletes exhibit abnormally low blood pressure at rest, and normal blood pressure during exercise. In contrast, accelerated capillary senescence due to obesity, smoking and toxic substances causes both essential hypertension and diabetes because it simultaneously increases microvascular flow resistance and undermines glucose uptake by cells. Thus, exercise conditioning is the most effective treatment for diabetes and hypertension.
The Three Pathways of Stress Mechanism Activation
Three synergistic pathways activate the stress mechanism and induce stress mechanism hyperactivity in accord with combinations of nervous activity and tissue disruption:
The nociception Pathway consists of peripheral nervous tissue disruption sensors called “nociceptors” that are in the skin and internal organs. These generate nervous activity called “nociception.” The nociception is conveyed via peripheral sensory nerves to specialized nociception pathways in the spinal cord. The spinal cord nociception pathways simultaneously transmit nociception to the brain and to sympathetic ganglia in the chest and abdomen. The brain interprets nociception as pain. The sympathetic ganglia generate sympathetic nervous activity that releases von Willebrand Factor from the vascular endothelium to close the capillary gate and inhibit organ function. Analgesia (narcotics and nerve blocks) inhibits the nociception pathway.
The cognitive pathway consists of consciousness and emotional mechanisms that assess sensory information (sight, sound, smell, touch, taste, nociception, etc.) for evidence of environmental danger and generates fear and anxiety that activates sympathetic nervous activity to facilitate “fight or flight.” Consciousness interprets nociception as pain, and it regulates nociception via inhibitory descending nervous pathways from the brain to the spinal cord. General anesthesia and sedatives inhibit the cognitive pathway. Its operation was discussed in greater detail in the previous issue of “Contentment” magazine.15
The tissue disruption pathway exposes tissue factor in extravascular tissues to blood enzymes in accord with tissue damage that disrupts the vascular endothelium. Tissue factor activates the enzymatic interaction to initiate tissue repair. Excessive tissue disruption releases tissue factor into systemic circulation, harmfully elevates thrombin generation, and causes systemic inflammation that disrupts organ function. Intravenous magnesium sulphate, EDTA, or trisodium citrate can inhibit thrombin activity, but stress theory suggests that a better treatment would be a drug that neutralizes tissue factor in systemic circulation.14
The Nature of Disease
Normally the stress mechanism functions quietly and efficiently, but like any mechanism, it has its limits. Excessive and unremitting combinations of tissue damage, pain, fear, anxiety, toxic substances, burns, surgery, microbial infestation, parasitism, excessive radiation, and other environmental stresses can induce harmful stress mechanism hyperactivity that consumes, wastes, and depletes stress mechanism substrates and enzymes, and produces excessive and defective quantities of soluble fibrin, insoluble fibrin, and thrombin, causing fever, fatigue, cachexia, malaise, inflammation, rashes, pustules, exudates, sclerosis, scars, and other disease manifestations. Such stress mechanism hyperactivity disrupts organ function and undermines its ability to resist environmental stress. Therefore, measures that alleviate stress mechanism hyperactivity and restore organ function can enhance antibiotic potency and penetration, optimize tissue repair, rid the body of infectious agents and toxic substances, halt destructive disease effects, and restore health.
Controlling Disease
Ideally all three stress mechanism activation pathways must be normalized simultaneously to optimize organ function and cure disease, but this is easier said than done, because each of the three pathways requires a separate treatment, and all medical treatments threaten toxicity, side effects, and risk. Furthermore, stress mechanism function is essential for life, so that excessive treatment is life-threatening.
Conclusion
This essay has provided a brief introduction to the stress mechanism that will serve as background information for subsequent essays that explain the unified theory of medicine postulated by Dr. Hans Selye, how it confers a unified theory of biology, and how its extended ramifications reside in the realm of science fiction. Those who seek greater detail may download copies of my published papers from my website18 or purchase my recently published book on Amazon.19
References
- Selye, H. A Syndrome Produced by Diverse Nocuous Agents. Nature 138, 1 (1936).
- Selye, H. The general-adaptation-syndrome and the diseases of adaptation. South Med Surg 113, 315-323 (1951).
- Selye, H. The general adaptation syndrome as a basis for a unified theory of medicine. Oral Surg Oral Med Oral Pathol 5, 408-413 (1952).
- Selye, H. Sketch for a unified theory of medicine. Int Rec Med Gen Pract Clin 167, 181-203 (1954).
- Peacock, E. E. Wound Repair. Third Edition edn, (W.B. Saunders Company, 1984).
- Tannenberg, J. Uber die Kapillartatigkeit. Verh dtsch. path. , 20:374 (1925).
- Bernard, C. An introduction to the study of experimental medicine. (Schuman, 1949).
- Henderson, Y. in Cyclopedia of Medicine (1940).
- Henderson, Y. A Lecture ON RESPIRATION IN ANAESTHESIA: CONTROL BY CARBON DIOXIDE. Br Med J 2, 1170-1175 (1925).
- Harvey, T. C. et al. Effect of carbon dioxide in acute mountain sickness: a rediscovery. Lancet 2, 639-641, doi:10.1016/s0140-6736(88)90465-5 (1988).
- Coleman, L. S. Four Forgotten Giants of Anesthesia History. Journal of Anesthesia and Surgery 3, 1-17, doi:10.15436/2377-1364.16.468 (2015).
- Hofman, W. I. Suicidal carbon dioxide asphyxiation: Case report. Mil Med 141, 477 (1976).
- Permentier, K., Vercammen, S., Soetaert, S. & Schellemans, C. Carbon dioxide poisoning: a literature review of an often forgotten cause of intoxication in the emergency department. Int J Emerg Med 10, 14, doi:10.1186/s12245-017-0142-y (2017).
- Welty-Wolf, K. E. et al. Coagulation blockade prevents sepsis-induced respiratory and renal failure in baboons. American journal of respiratory and critical care medicine 164, 1988-1996 (2001).
- https://www.stress.org/contentment-magazine-winter-2021-2.
- https://www.osha.gov/publications/hib19960605
- Campbell, A. & Poulton, E. P. Oxygen and carbon dioxide therapy. (Oxford University Press, H. Milford, 1934).
- stressmechanism.com
- https://www.amazon.com/Years-Lost-Medical-Advance-discovery/dp/0578822601/ref=sr_1_1?crid=2Y4YZ3672B2VV&keywords=Hans+Selye+lewis+coleman&qid=1642176982&sprefix=hans%2520selye%2520lewis%2520coleman%2Caps%2C177&sr=8-1
ABOUT THE AUTHOR

Lewis Coleman, MD, FAIS is a board-certified anesthesiologist who completed his BS degree in biology at Ohio State University, earned his MD degree from New York Medical College, and completed his surgical internship and anesthesiology residency at UCLA, followed by 40 years in private practice. Coleman’s basic sciences instruction at NYMC miraculously coincided with the two-year sojourn of Dr. Johannes Rhodin, a famous Swedish pioneer of electron microscopy who was retained by the school to upgrade its curriculum. Dr. Rhodin was an expert on the stress theory of Hans Selye. His stress theory lectures devastated the dogma of classical physiology and convinced Coleman that stress theory represented the future of medicine. Many years later, these lectures miraculously enabled Coleman to identify Selye’s long-sought stress mechanism. Thus identified, the stress mechanism enables Selye’s “Unified Theory of Medicine” that promises a new era of health, longevity, and freedom from the eternal curse of disease. Its implications exceed the bounds of medicine and confer a “unified theory of biology” that explains embryology, extinction, evolution, ethology, intelligence, anatomy, taxonomy, the Cambrian explosion, and dinosaurs, and resolves the disparities of Darwin, Lamarck, Baldwin, and saltation. Its distant implications reside in the realm of science fiction. His website http://www.stressmechanism.com is dedicated to stress theory and offers relevant materials free of charge. His book, 50 Years Lost in Medical Advance: The Discovery of Hans Selye’s Stress Mechanism, is available on Amazon.
Contentment Magazine
The dictionary defines “content” as being in a state of peaceful happiness. The AIS magazine is called Contentment because we want all of our guests and members to find contentment in their lives by learning about stress management and finding what works best for each them. Stress is unavoidable, and comes in many shapes and sizes that makes being in a state of peaceful happiness seem like a very lofty goal. But happiness is easy to find once you are able to find ways to manage your stress and keep a healthy perspective when going though difficult times in life. You will always have stress, but stress does not always have you!

Leave A Comment
You must be logged in to post a comment.