The use of cranial electrotherapy stimulation in the treatment of multiple sclerosis
Smith, Ray B. The use of cranial electrotherapy stimulation in the treatment of multiple sclerosis. The Original Internist, 9(3):25-28, September, 2002. Download Article.
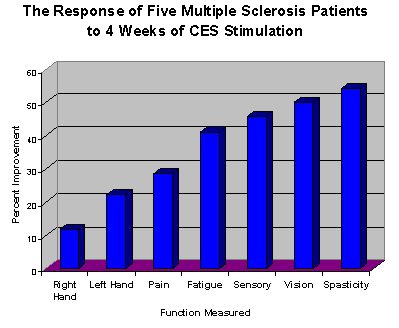
When the author worked as vice president of research at Electromedical Products International, Inc. he received 2 testimonial letters giving glowing reports of the effectiveness of Alpha-Stim CES in reducing or alleviating several of the major symptoms of multiple sclerosis (MS). Until then no one had considered the possibility of CES as a treatment for MS. The author then checked through the EPI database of thousands of warranty cards and found 12 cards that had been sent in by patients who had been prescribed an Alpha-Stim for their MS. They claimed an average improvement of 46.57% (range of 0-99%) in their symptoms. 10 of the 12 were females, ages ranged from 21-52 year old (average age was 39), and they used CES 1 – 8 weeks (average 4.4 weeks, or one month). Based on this preliminary evidence a pilot study was conducted. After giving informed consent, 5 patients (3 females) used the Alpha-Stim for one hour a day for one month, at 0.5 Hz and a comfortable level of current between 100 and 300 µA. MS diagnoses ranged from 10 – 38 years (average of 21), ages ranged from 53 – 68 years old (average age was 60). Improvement was noted in 7 of the 10 symptoms measured. Self rated spasticity improved 54%, vision improved 50%, sensory (ability to feel) improved 45%, fatigue improved 40%, pain improved 29% and hand function improved 22% in the left hand and 12% in the right hand. Bladder, cognitive and mobility/gait function was not rated as significantly improved by these patients. The author concluded that while this is only a pilot study, the results were consistent with letters and warranty cards received and so this study is probably a fairly accurate assessment of what a physician could expect when prescribing a CES device for use by MS patients.